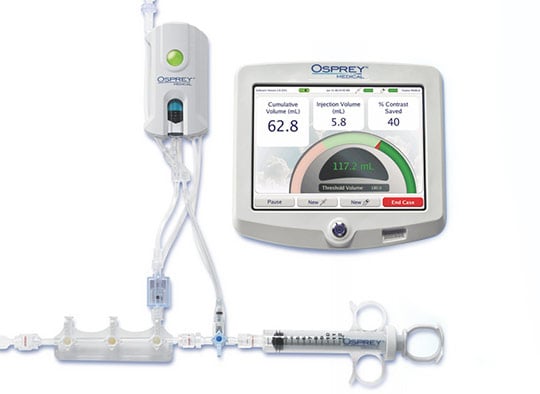
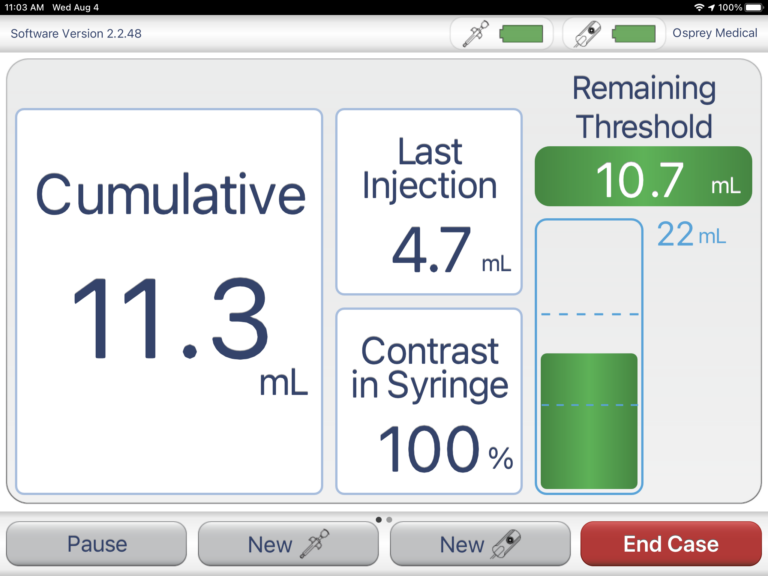
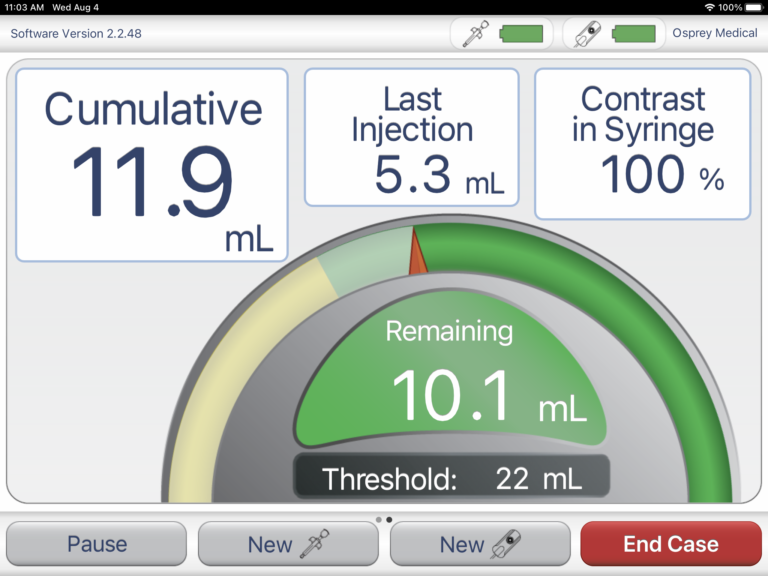
Though Osprey Medical has been driving innovation in the world of angiography, the company has a fairly small engineering team with limited expertise in connected software product development. When Osprey first approached Punch Through, its legacy device was a costly, custom touchscreen tablet. The legacy device required hand-delivered manual software upgrades and often time-consuming maintenance — as it was sometimes difficult to isolate and debug issues with the system.
First looking to resolve connectivity problems and improve the overall reliability of its device, then looking to design a next-generation product, Osprey Medical brought in Punch Through to architect and develop a solution due to our immense connected product development experience.